There are four bonding pairs of electrons and no lone pair of electrons in this molecule. So in the Lewis structure of CH 4 or Methane, there are four single or covalent bonds between each Hydrogen and Carbon atom. Each bond requires two valence electrons, and hence eight valence electrons are used up by forming bonds. 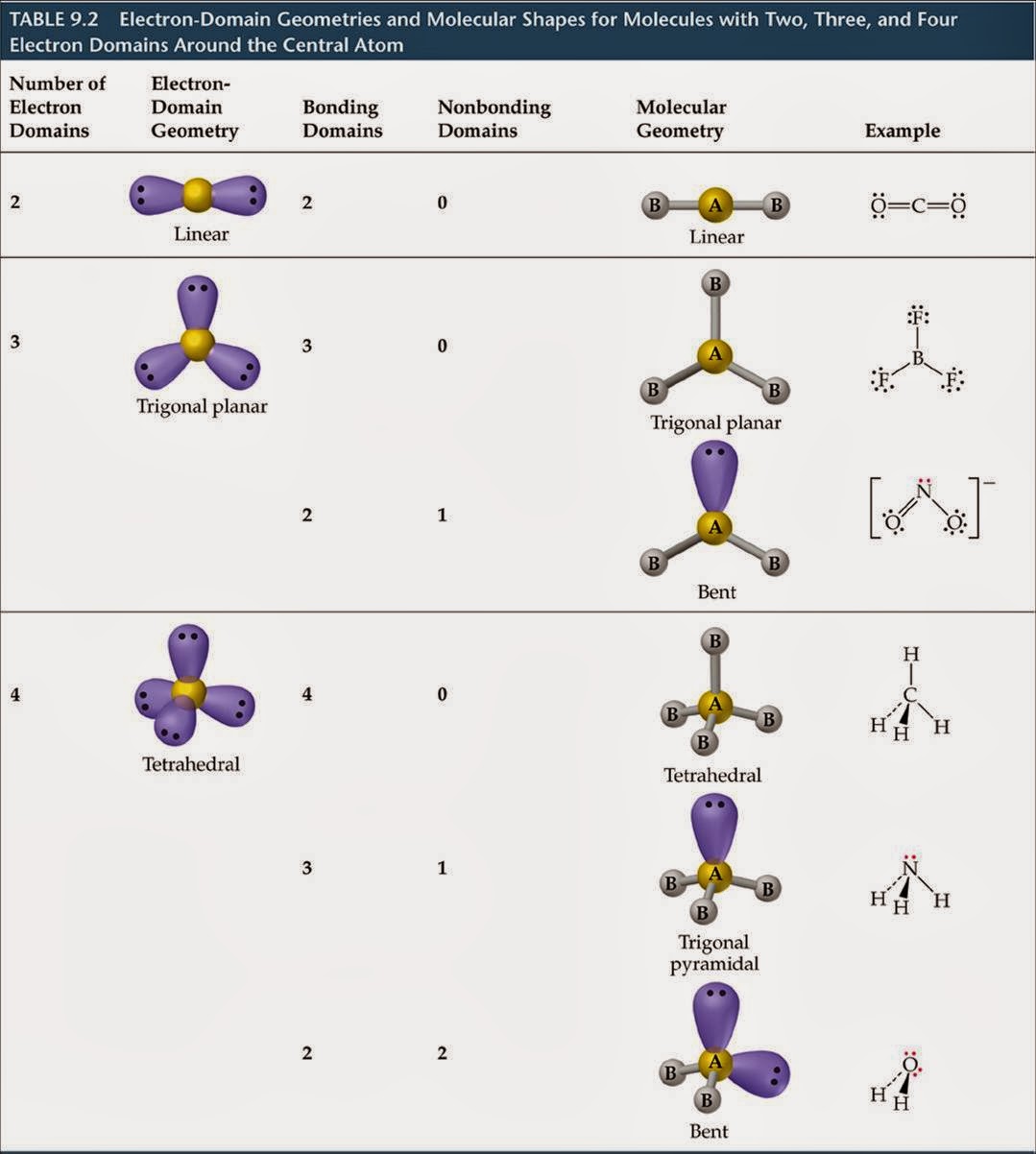
So for doing that, it will share one valence electron of the Carbon atom.Īs Carbon has four valence electrons, it will share all four electrons with the Hydrogen atoms. Now each Hydrogen just needs one more valence electron to attain a stable structure. Arrange all the Hydrogen atoms around the Carbon atom. Let us look at the Lewis Structure of CH 4 and determine how the atoms are arranged in the molecule.Ĭarbon in Methane takes the central position as it is less electronegative than the Hydrogen atoms. However, many elements are exceptions to this rule. Lewis structures are based on the octet rule, which says an atom must have eight valence electrons in its outer shell to attain a structure similar to the closest noble gas. Lewis structure is also referred to as electron dot structure.ĭots are represented to show the electrons, and lines are used to indicate the bonds between the atoms. The electrons that participate in bond formation are called the bonding pair of electrons, while those that don’t are known as nonbonding pairs of electrons. Lewis structure is the pictorial representation of the arrangement of valence shell electrons in the molecule, which helps us understand the atoms’ bond formations. 
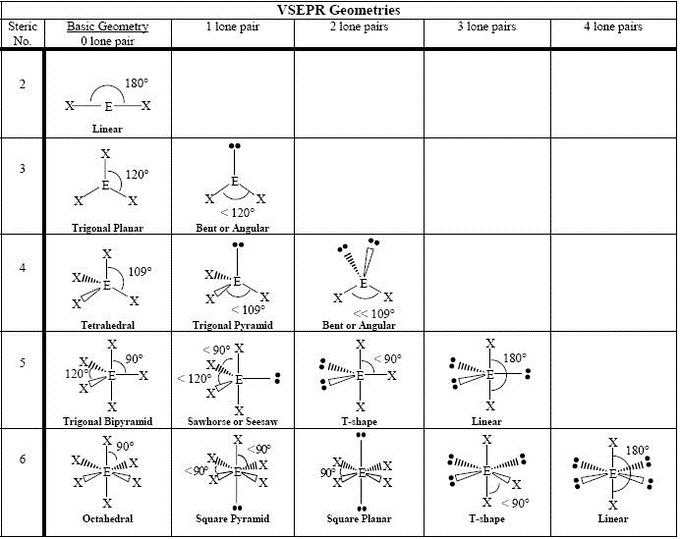
Thus there eight valence electrons for Methane. The total number of valence electrons in CH4 adds up to be 8 (4 in carbon and 1 from each hydrogen atom). This gives us a total of 4 valence electrons in carbon and 1 in Hydrogen. The electrons present in the outermost shell of an atom are termed as valence electrons. So, in Carbon, the electronic configuration will be 1s2 2s2 2p2 and the electronic configuration of Hydrogen will be 1s1. Hydrogen is the very first element in the periodic table having an atomic number 1. The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.The electrons that participate in the bond formation and are in the outermost shell of the atom are referred to as valence electrons.Ĭarbon is a p block element present in group 14th and 2nd period in the periodic table and has atomic number of 6. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion.The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral.Molecular geometries take into account the number of atoms and the number of lone pair electrons.Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
